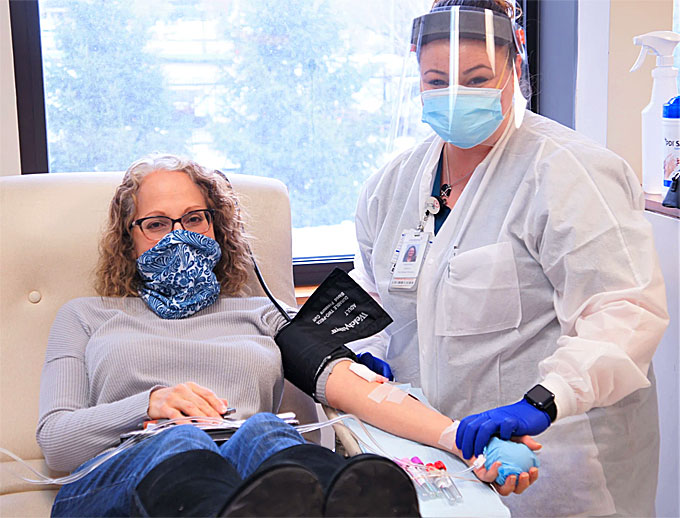
Working in conjunction with hospital partners Vitalant has launched a program to treat COVID-19 patients with blood plasma donated by individuals who have recovered from the disease.
This blood component, known as “convalescent plasma,” contains antibodies that may provide seriously ill patients an extra boost in fighting the disease.
There currently are neither vaccines nor proven treatments for COVID-19, because the virus is so new.
Although trials for a vaccine are underway, it is expected to be months before one is approved.
(People who are sick or injured need blood to survive. Please schedule a blood plasma donation today. Courtesy of Vitalant and YouTube.)
The U.S. Food and Drug Administration (FDA) has identified and approved convalescent plasma treatment as an “emergency investigational new drug.”
It is currently the only antibody treatment available to COVID-19 patients and, as such, is a promising new tool.
Vitalant is collecting convalescent plasma by appointment only at its locations in Montvale, Paramus, and Parsippany, NJ.
Those interested in donating plasma are encouraged to apply through the Vitalant website Vitalant.org/covidfree.
Additional details are available by calling 866-CV-PLSMA (866-287-5762).
“This is an example of the medical and research communities coming together to effect real change during this pandemic,” said Maria De Los Angeles Muñiz, MD, Medical Director at Vitalant’s Montvale, NJ location.
“Convalescent plasma is a promising response tool for fighting COVID-19 in patients, and potentially for those working on the front lines.”
Eligibility criteria for convalescent plasma donors are:

-
Prior diagnosis of COVID-19, documented by a laboratory test
-
Complete resolution of symptoms for at least 14 days
-
Meet all other current FDA donor eligibility requirements to donate plasma
Even for those who meet these requirements, additional tests may be required.
In some instances, those who have recovered from COVID-19 may be able to donate, even if they did not have an initial laboratory test.
The blood collection process is safe.
In addition to the vigorous FDA-mandated testing of all blood components donated, Vitalant staff follow rigorous safety and disinfection protocols at its blood drives and donation centers.
Finally, giving blood has no impact on the donor’s immune system.

“Sick hospital patients rely on blood donors—there is no other source for blood,” Vassallo said.
“We need healthy individuals to donate blood, while following guidance from the CDC and other agencies.”
COVID-19 transmission occurs like the spread of the flu (influenza).
And although there is currently no vaccine for COVID-19, to reduce the risk of infection, the CDC recommends washing hands often with soap and water for at least 20 seconds, and avoiding touching the eyes, nose, or mouth with unwashed hands.
Vitalant is closely monitoring the situation and will quickly implement any necessary changes as new information emerges from the CDC, FDA and AABB.
Every day, almost 5,000 blood donations are needed to meet the needs of people throughout the country, and Vitalant’s 800,000 donors supply 1.8 million donations a year.
 Vitalant is the nation’s second largest community blood service provider, supplying comprehensive transfusion medicine services for nearly 1,000 hospitals and health care partners for patients in need across 40 states.
Vitalant is the nation’s second largest community blood service provider, supplying comprehensive transfusion medicine services for nearly 1,000 hospitals and health care partners for patients in need across 40 states.
In addition to blood products, Vitalant offers customers transfusion services, medical consulting, quality guidance, ongoing education, research and more.
For more information and to schedule a donation, please visit vitalant.org or call 877-25-VITAL (877-258-4825).
You can also donate at the Red Cross, go to https://www.redcrossblood.org/give.html/ or call 1-800-RED CROSS (1-800-733-2767).
AST strives to meet a 3 STAR trustworthiness rating, based on the following criteria:
- Provides named sources
- Reported by more than one notable outlet
- Includes supporting video, direct statements, or photos
Subscribe to the AST Daily News Alert Here.
Learn More…
Healthy Blood Donors Are Urgently Needed for Hospital Patients Today